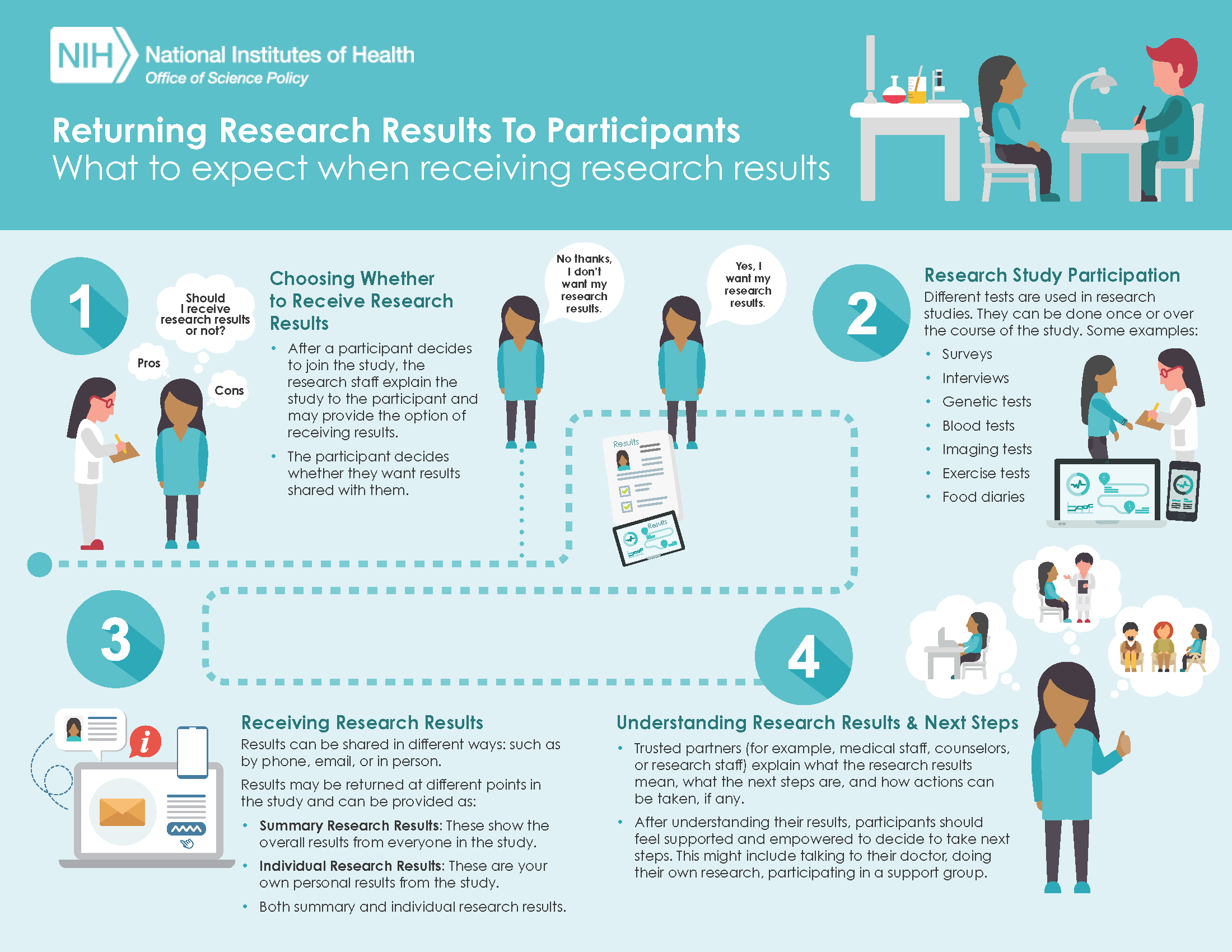
Return of Research Results
NIH is committed to making the return of research results to participants in NIH-funded clinical research standard practice. Returning research results to study participants enhances transparency, builds trust, shows respect for their contributions, and engages them as partners in research. OSP is leading the development of policies and supporting guidance materials to enable the research community to return research results responsibly to participants.
Returning Research Results
Research participants are essential to NIH achieving its mission. Building partnerships with participants enables NIH to foster innovative health discoveries and expand medical knowledge. NIH has long supported engaging participants in informing scientific priorities and study design to ensure that research in meaningful to them and society. NIH is committed to furthering engagement with participants by promoting and enabling return of research results.
Building from a multi-year effort involving the community and public conversations, OSP is taking a key step to implementing NIH’s roadmap for engaging the public as partners in research by employing strategies that return value to participants. Participants want and expect the opportunity to receive both information on the overall study findings as well as information that is specific to them, in return for their contributions to science.
What’s Next: Making Return of Results Standard Practice
While NIH has some programs actively engaged in returning research results to participants, it does not occur consistently in practice across the clinical research enterprise. To address this inconsistency, NIH is working with the community to develop a policy framework to make the responsible return of research results standard practice.
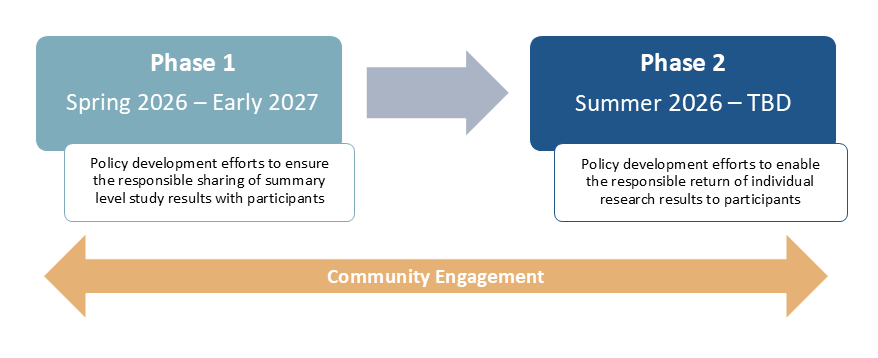
OSP is embarking on a two-phase policy development process that will directly engage the community.
- Phase 1: This phase aims to build upon previous efforts and existing best practices to develop a policy to ensure the responsible sharing of summary level study results with participants (Spring 2026 – Early 2027). More information on Phase 1 can be found here.
- Phase 2: Informed by the outcomes and lessons from Phase 1, this phase aims to develop a policy to enable the responsible return of individual research results directly to participants (Summer 2026 – TBD).
Policy on Sharing Summary Level Study Results with Participants
Summary level study results, also commonly known as aggregate research results, are concise overviews of a study, that include objectives, methodology, and cumulative findings. These summary level results are presented in plain language that is understandable, meaningful, accessible, and appropriate. Sharing these results is a recognized best practice that enhances transparency, demonstrates respect for participants’ contributions, and fosters trust in clinical research. By responsibly sharing study results in plain language, researchers can strengthen relationships and partnerships with research participants.
Policy Development Process
Starting in Spring 2026, NIH will undertake a comprehensive policy development process to establish requirements for researchers and institutions to responsibly share summary level study results with research participants in plain language for all NIH-funded clinical research. A timeline for this process is presented below.

Stakeholder Engagement and Opportunities for Public Input
NIH is committed to working with the public to inform a draft policy on the sharing of summary level study results with participants. Public input is essential to developing a robust policy that reflects various stakeholder perspectives.
Individuals are encouraged to share ideas and feedback at any time here. Individuals may provide comments on any aspects of sharing summary level study results, and all comments will be considered by NIH. Specific topics of interest have been highlighted, and this mechanism will remain open until the draft policy is released. Members of the public will also have the opportunity at that time to provide feedback on the draft policy.
In addition to on-demand commenting, NIH will be hosting a webinar to raise awareness of the policy development process and to obtain public input. The target audience for this webinar is research participants, patients, caregivers, researchers (particularly clinical researchers), patient advocates, clinicians, Institutional Review Board and Human Research Protection Program members and administrators, and members of the general public. More information on the webinar can be found by clicking on the flyer below.
Provide Feedback to Inform a Draft Policy for Sharing Summary Level Study Results
Do you have ideas for the development of a draft NIH policy for the sharing of summary level study results with participants? We want to hear from you!
Starting in Spring 2026, NIH is undertaking a comprehensive policy development process that will establish requirements for researchers and institutions to responsibly return summary level study results to research participants in plain language for all NIH-funded clinical research. Public input is vital to ensuring NIH develops a policy that is flexible, forward thinking and serves the best interest of the research participants and the researcher community.
NIH wants to ensure everyone can make their voice heard on this important issue. You can share your views by submitting comments to OSP at any time by using this portal.
Your general input on this topic is welcome and appreciated. NIH will consider all comments received. Of note, NIH is particularly interested in hearing individual comments on:
What to include in summary level study results shared with participants. Specifically:
- Elements and information that should be expected to be included in summary level study results shared with participants
- Situations when not sharing summary level study results with participants may be justifiable
- Strategies for generating plain language summaries, including the use of AI tools
How to share summary level study results with participants. Specifically:
- Best practices for participant engagement to ensure that results are returned in meaningful ways and participants know what to expect
- Acceptable and preferred methods for sharing and receiving summary level results
- Potential costs related to generating and sharing summary level results
- Additional considerations related to ensuring results are returned responsibly
When to share summary level study results with participants. Specifically:
- Timing of sharing summary level study results (during or after the study)
Please submit your thoughts below. Commenters can include their names and email addresses, or submit anonymously.
Returning Individual Research Results to Participants
Individual research results include participant-specific findings, such as personal lab results or health information. These are different from summary level study results, which provide an overview of collective study findings. Individual research results offer participants information about their own data generated through their participation in research. Returning individual research results can support participants in understanding their personal information and may inform their own health decisions. Additionally, returning individual research results strengthens partnerships between the researcher community and participants by considering participants as partners in advancing scientific knowledge.
A National Academies of Sciences, Engineering, and Medicine (NASEM) report Returning Individual Research Results to Participants, co-sponsored by NIH, examined evidence on the benefits, harms, and costs of returning individual research results. It also considered the ethical, societal, regulatory, and operational issues related to the return of an individual’s research results in the context of human biospecimens research. As a result of this study, various NIH programs and initiatives have moved towards a more participatory model of returning individual research results to participants. However, this occurs inconsistently across clinical research generally.
NIH is committed to working with the public to enable an environment that is appropriate and conducive to allowing greater return of individual research results. More information is coming soon about the policy development process and engagement opportunities related to returning individual research results.
Developing Evidence to Enable Responsible Return: OSP/NIEHS/NHGRI Effort
In 2024, OSP, in partnership with the National Institute of Environmental Health Sciences (NIEHS) and the National Human Genome Research Institute (NHGRI), hosted a kickoff meeting for recipients of NIH grants focused on charting a responsible path forward for reporting back environmental health and non-genomic research results. Researchers from across the country presented on their awarded projects, all aimed at informing and developing guidelines, educational resources, and community engagement approaches for effective communication of research results.
Select Other NIH Efforts to Promote Responsible Return of Research Results
- All of Us Research Program
- Environmental influences on Childhood Health Outcomes (ECHO) Program
- National Human Genome Research Institute
- National Institute of Diabetes and Digestive and Kidney Diseases
Infographics for Communicating About Return of Research Results
OSP created infographics to help communicate the concepts of returning research results. The first infographic displays what research participants can expect when participating in a study that provides them with the option to receive their research results. (click to enlarge)
The second infographic displays key considerations for researchers to think about when designing and conducting studies that plan to return research results to participants. These visual resources are intended to be used to help communicate these concepts in a way that’s more easily understood. (click to enlarge)